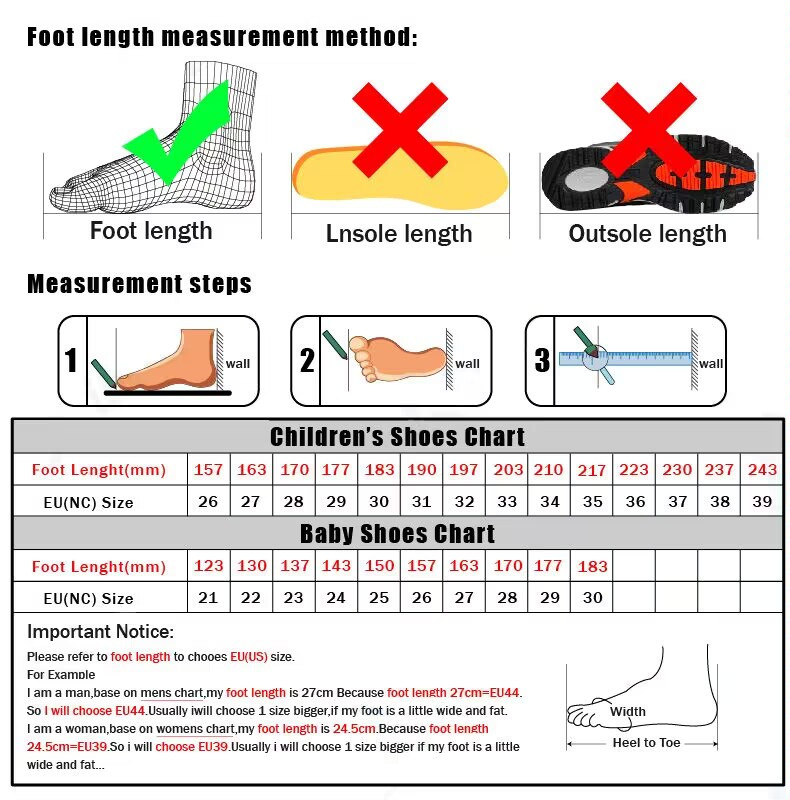
Παιδικά παπούτσια ποδοσφαίρου TF Μποτάκια ποδοσφαίρου κατά της σύγκρουσης Αντιολισθητικά Αντιολισθητικά Ανθεκτικά Απορροφητικά κραδασμών - Badu.gr

Αγοράστε Νέα Plus Velvet Thicken Γυναικεία Αθλητικά Παπούτσια Μόδα Casual Ζεστά Παπούτσια Αντιολισθητικά και απορροφητικά τους κραδασμούς Χειμερινά παπούτσια για τρέξιμο | Joom

Παιδικά παπούτσια ποδοσφαίρου TF Μποτάκια ποδοσφαίρου κατά της σύγκρουσης Αντιολισθητικά Αντιολισθητικά Ανθεκτικά Απορροφητικά κραδασμών - Badu.gr

Adidas Ultraboost 22 GX3062 Ανδρικά Αθλητικά Παπούτσια Running Core Black / Cloud White | Skroutz.gr

Αγοράστε Νέα Plus Velvet Thicken Γυναικεία Αθλητικά Παπούτσια Μόδα Casual Ζεστά Παπούτσια Αντιολισθητικά και απορροφητικά τους κραδασμούς Χειμερινά παπούτσια για τρέξιμο | Joom

Παιδικά παπούτσια ποδοσφαίρου TF Μποτάκια ποδοσφαίρου κατά της σύγκρουσης Αντιολισθητικά Αντιολισθητικά Ανθεκτικά Απορροφητικά κραδασμών - Badu.gr

Πατάκια Απορροφητικά Σκόνης 2 τεμ. Ορθογώνια Μπλε 90x150 εκ.,Χαλάκια Πόρτας,Διακόσμηση,Το συγκεκριμένο πατάκι απομακρύνει τη βρομιά και την υγρασία από τα παπούτσια σας, διατηρώντας καθαρές τις επιφάνειες των εσωτερικών σας χώρων












