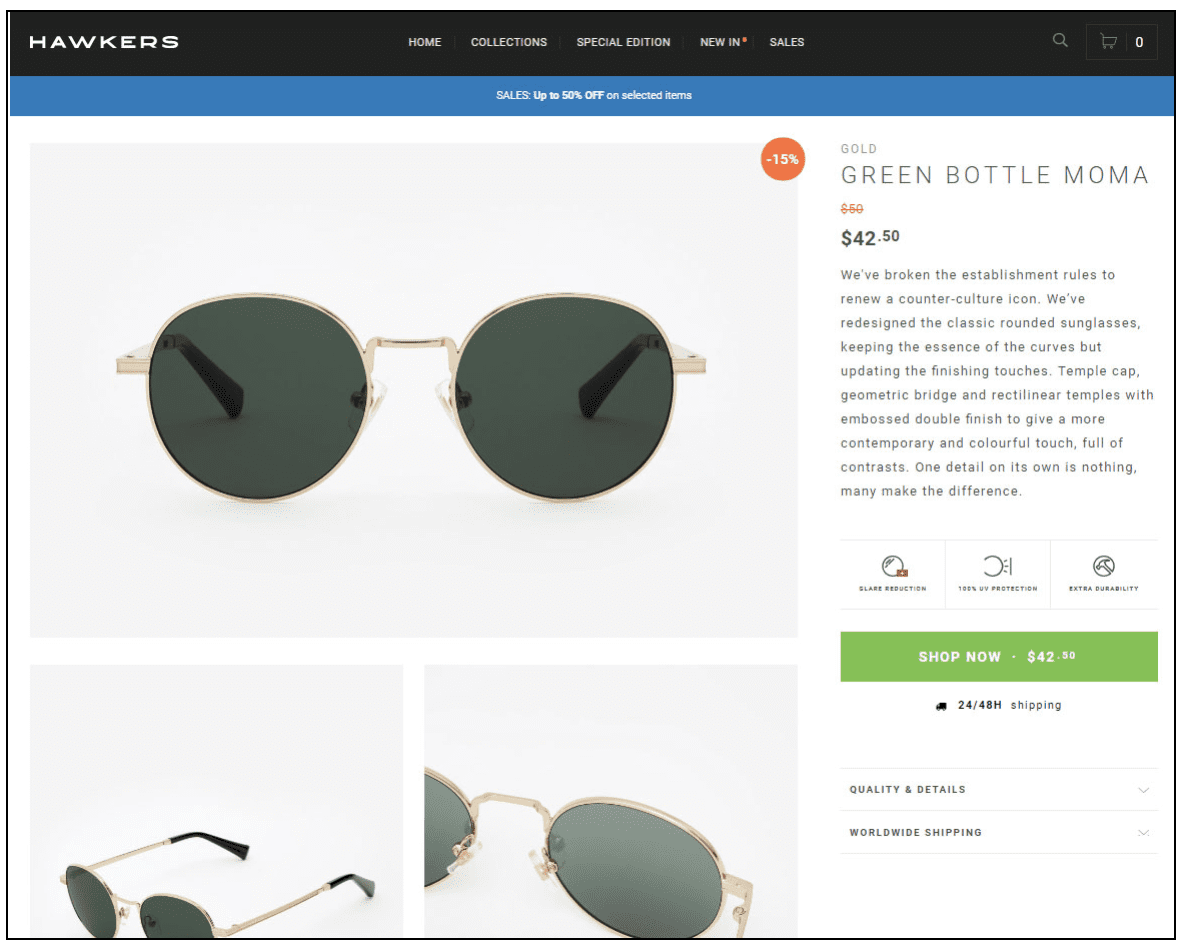
Hawkers Gold Green Bottle MOMA Unisex Sunglasses, TR18 UV400, 46: Buy Online at Best Price in UAE - Amazon.ae

HAWKERS | Gold Green Bottle MOMA Sunglasses for Men and Women, Unisex. UV400 Protection. Official product designed in Spain MOMA2 | 顏色: 綠色| HKTVmall 香港最大網購平台

Hawkers Gold Green Bottle MOMA Unisex Sunglasses, TR18 UV400, 46: Buy Online at Best Price in UAE - Amazon.ae

Hawkers Sunglasses Gold Green Bottle Moma For Men, Women, Unisex. Proteccion Uv400. Official Product Designed In Spain - Sunglasses - AliExpress

Hawkers Unisex Gold Green Bottle Sunglasses, Gold Green Bottle, 50 US : Amazon.com.au: Clothing, Shoes & Accessories