BLE S/2 ΣΚΟΥΛΑΡΙΚΙΑ ΣΕ ΛΕΥΚΟ/ΧΡΥΣΟ ΧΡΩΜΑ ''ΚΟΡΑΛΙΑ'' ΜΕ ΧΑΝΤΡΕΣ 4X1X5 Χρυσό,Λευκό-Ιβουάρ Σίδερο Συνθετικό Γυαλί

Βοηθητικό Τραπεζάκι Pino Megapap Μεταλλικό - Mdf Χρώμα Λευκό - Χρυσό 35X35X58Εκ. - Megapap | SHOPFLIX.gr

Cyclop Tk Lighting Industrial Μεταλλικό Φωτιστικό Δαπέδου με Τρία Στρόγγυλα Καπέλα σε 2 Χρώματα 163Υ - FloBaLi

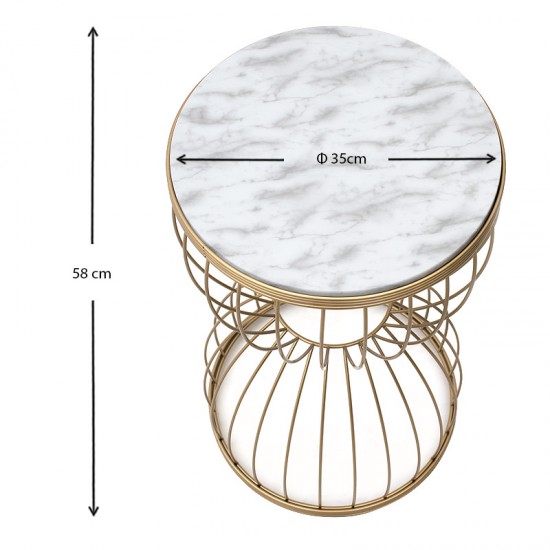
Βοηθητικό τραπεζάκι Hoffman Megapap μεταλλικό - Mdf χρώμα χρυσό - λευκό εφέ μαρμάρου 35x35x58εκ. - YouBath.gr

ΜΕΤΑΛΛΙΚΟ ΔΙΑΚΟΣΜΗΤΙΚΟ ΤΟΙΧΟΥ ΜΕ ΚΑΘΡΕΦΤΗ Fylliana 3767 ΜΑΥΡΟ-ΛΕΥΚΟ-ΧΡΥΣΟ ΧΡΩΜΑ 98x5x63εκ | Διακοσμητικά τοίχου | Fylliana